Analysis Of Silver Mercury And Lead Lab - Qualitative analysis is carried out through the reactions which are easily perceptible to our senses such as sight and smell. Analytical techniques for analysis of heavy metals by:
Pin On Revision
When choosing the analytical technique, a lot of factors needs to be looked at such as cost, sensitivity.

Analysis of silver mercury and lead lab. Qualitative analysis are easily confused, and it is therefore very important that you be diligent about labeling your various test tubes, and that you record your observations immediately. Qualitative analysis is a branch of analytical chemistry that identifies particular substances in a given sample of material. In such an analysis, there are two distinct phases.
Then, a different test is performed to uniquely confirm the identity of each separated ion. Unknown hcl crystals white ppt ions of group i Exposure to metallic mercury vapors at higher levels for shorter periods of time can lead to lung damage, vomiting, diarrhea, nausea, skin rashes, increased heart rate or blood pressure.
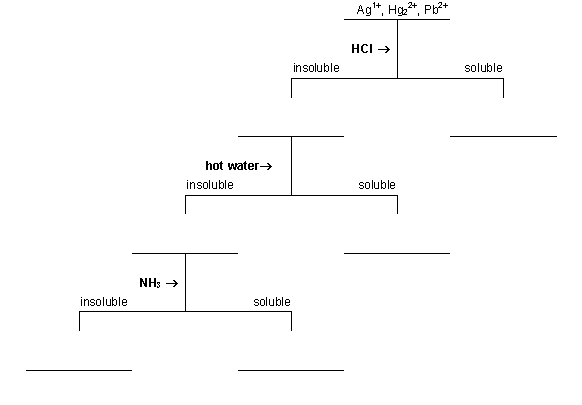
However, for direct analysis of drinking water (turbidity mercury</strong> in solution can be determined by direct analysis pneumatic nebulization provided gold is. In this lab, we develop a scheme to separate and identify the silver ion (ag +), the mercury(i) ion (hg 2 2+), and the lead(ii) ion (pb 2+). Write a balanced net ionic equation for each of the following reactions:
Start studying analysis of silver, mercury, and lead (quiz 4). Chemical analysis depending upon the nature of ore samples and radicals/elements to be analyzed. Solubility product constants indicate that lead (ll) chloride is much more soluble than the chlorides of mercury (i) or silver (i).
In macro analysis, 0.1 to 0.5 g of substance and about 20 ml of solution is used. The choice of any analytical procedure or instrumental analytical techniques also depends on the concentration of elements in parts per million (ppm) or otherwise. Lab techniques • ensure all glassware is clean, but it does not need to be dry.
Analysis of silver, mercury, and lead (quiz 4) flashcards | quizlet. The washing water that we examine is added with some drops of silver nitrate (agno 3). Because the solubility in water of pbcl 2 increases from about 0.036 mol/l at 20°c to about 0.12 mol/l at 100°c, cold solutions are used in this procedure to maximize precipitation of the lead…
Thus each procedure of chemical analysis has its own merits and demerits and selection Most human exposure to mercury is caused by outgassing of mercury from dental amalgam, ingestion of contaminated fish, or occupational exposure, according to the world health organization [7, 8].mercury exists in nature primarily as elemental mercury or as a sulfide and is found in the earth's crust at approximately 0.5 parts per million. The aim of this case study is to choose a suitable instrumental analytical technique to investigate the concentration of heavy metals in sludges collected from contaminated water courses.
Step substance reagent result inference 1. Laboratory techniques used in qualitative analysis. Mercury is unique in that the elemental form cf this metal has appreciable vapour pressure at room temperature, and it is therefore possible to determine
First, a scheme must be developed to separate the ions from each other. • use distilled water at all times • label test tubes to avoid confusion • mix solutions by flicking the test tube • estimate volumes by assuming 20 drops by pipette ≈1ml 1) treatment with sodium acetate (ch3coona) and potassium chromate (k2cro4) the aim of this assay is the precipitation of pbcro4.
The precipatation of the chloride of hg2 2+ in step 1. Replaced by sophisticated instrumental methods for practical analysis. In this experiment, you will analyze a known solution that contains all the group i cations—silver, lead, and mercury(i)—and an unknown solution to determine which of these ions are present and which are absent.
In this experiment, you will analyze a known solution that contains all the group i cations—silver, lead (ii), and mercury(i)—and an unknown solution to determine which ions are present and which are absent. Once separated silver and mercury (i) from pb2+ we can proceed with its identification. Give the formula and colors for the chlorides formed with silver, lead and mercury(i).
+the chlorides of ag , pb 2+, and hg 2 are all insoluble in cold water. Methods the detection limit for mercury is about 200 yg/fl. When hg 2 cl 2 is treated with aqueous nh 3 a reaction occurs in which free mercury and amidochloromercury(ii) are formed.
For semimicro analysis, 0.05 g substance and 1 ml solution is needed while for micro analysis amount required is very small. Hg 2 cl 2 (s) + nh 3 (aq) → hg(l) + hgnh 2 cl(s) + hcl(aq) the hgnh 2 cl is a white solid, while the hg in a finely divided state appears black. Learn vocabulary, terms, and more with flashcards, games, and other study tools.
To begin the study of the first group of three cations, ag+, pb2+, and hg 2 2+ ions, which are separated out as insoluble chloride salts. Increased exposure of mercury can alter brain functions and lead to shyness, tremors, memory problems, irritability, and changes in vision or hearing. Mercury(i) ions exist as dimers in aqueous.
It is suggested that you use the following format for your lab notebook: In water analysis, the need exists for routine mercury determina tions down to levels of below 1 yg/s.
Freddie Mercury Poster By Wirdou Freddie Mercury Nerd Humor Mercury
Solved Ag Group Aim Identification Of A Group Of Cations Cheggcom
Lab 4 - Qualitative Analysis
Chem Review - Reactivity Series Chemistry Help Chemistry Lessons Science Chemistry
Thyrocare Aarogyam C Profile 63 Tests With Vitamin D And B12 Rs 1000 Lipid Profile Preventative Health Health Check
Analysis Of The Silver Group Cations
542 Reactivity Series And Extraction Of Metals - Spm Science Chemistry Basics Study Chemistry Teaching Chemistry
Chemical Properties Of Metals And Nonmetals - A Plus Topper Chemicalpropertiesofmetals Chemical Property School Study Tips Sixth Grade Science
Reactivity Series Of Metals Chemistry Education Science Chemistry Chemistry
Mountain View Chemical Reactions Chemistry Lessons Chemistry Classroom Chemistry Basics
Mobil 1 0w40 European Car Formula Voa Virgin Oil Analysis German Autohaus Chattanooga Tennessee Motul Analysis Blackstone
Alphabetical List Of The Elements - Five Js Homeschool Periodic Table Words Periodic Elements Periodic Table With Names
Haramain Urbanist Prive - Silver And Gold For Stylish Women Reshared From Al Haramain Perfumes Uk Perfume Uk Perfume Store Alcohol Free Fragrance
Sat-chemistry-the-laboratory-laboratory-safety-rules-311-1 Chemistry Safety Rules Laboratory